
Curriculum Vitae
- B.S. Chemistry, San Diego State University
- Ph.D. Chemistry, University of Colorado, Boulder, Advisor . Dr. W. A. Castleman
- Postdoc (Microwave Spectroscopy of van der Waals Complexes) with Dr. W. Klemperer; Harvard University
- Assistant and Associate Professor, Department of Chemistry, University of Rhode Island
Research Interests
My research interests have focused on the character of intermolecular interactions. Currently, I am involved in three separate projects in this area: 1) ligand exchange kinetics on the surface of silver nanoparticles . this work is done at SDSU in collaboration with Dr. David Pullman; 2) binding constants of sulfonamides with fluoride ion and acetate ion . this work is also done at SDSU in collaboration with Drs. Somanathan and Lafferty; and 3) microwave spectroscopic and theoretical studies of van der Waals complexes containing alkanes, e. g., CH4-C3H8 . this work is primarily done at Wesleyan University in the lab of Dr. Stew Novick. Further details of these projects are given below. A number of undergraduates have been involved in the first two projects, and I welcome further student involvement.
Research Summaries
1) Ligand exchange kinetics on the surface of silver nanoparticles. The motivation for this project is to better understand the properties of silver nanoparticles in aqueous environments, particularly those of biological interest. The surface composition of silver nanoparticles, which affects their behavior, depends on the concentration of the components in solution and their relative binding to the surface. We explore the binding competition by monitoring the changes in the surface plasmon resonance peak, which depends on the surface coverage and composition. Other methods we are using, or plan to use, are Surface Enhanced Raman Spectroscopy (SERS), which is useful for analyzing surface constituents, and Dynamic Light Scattering with zeta potential methods for determining the surface charge. We have completed work on halide-induced aggregation of nanoparticles (J. Phys. Chem. C, 116, 8305 (2012)), and are now investigating the details of the mechanism of chloride surface adsorption and aggregation through further kinetic experiments. We are also investigating the pH dependence of amino acid adsorption on silver nanoparticles.
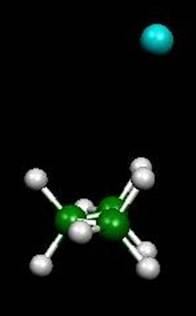
Vibrationally averaged
structure of Ar-propane
2) Binding constants of sulfonamides with fluoride ion and acetate ion. Fluoride and acetate ions complex strongly to the amine protons in sulfonamides. In molecules with multiple sulfonamide groups, this binding can cause distortions in the structure. We are analyzing complexes with a tetrakis sulfonamide that has been synthesized by Dr. Somanathan. By measuring and evaluating the proton shifts of the complexes, we can obtain binding constants and some information about changes in the sulfonamide structure. We are also carrying out Gaussian calculations on the complexes to help in this evaluation. To understand more about the fundamental effect of anion bonding to sulfonamides, we are also studying the much simpler benzene sulfonamide.
3) Microwave spectroscopic and theoretical studies of the van der Waals complex CH4-C3H8. Because of their low binding energy, relatively large size, and internal rotation complications, very few van der Waals complexes with alkanes have been studied. Yet, these studies can be very helpful in understanding interactions which, although weak, are ubiquitous, and therefore of importance in polymeric folding. CH4-C3H8 is perhaps the simplest alkane-alkane complex which has a large enough dipole moment for microwave absorption (~0.1 D). We have been able to measure, and partially analyze, the spectrum of CH4-C3H8, and its structure appears to be very similar to that of Ar-C3H8 (J. Chem. Phys. 127, 184306 (2007)). The next step is to do high-level ab initio calculations on the complex in order to completely work out the structural details and the evident internal motion of the methane subunit.
Selected Publications
- M. Gabriela Espinosa, Alexandra M. Mendoza, Malory L. Hinks, David Pullman, and Karen I. Peterson, .Kinetics of Halide-Induced Decomposition and Aggregation of Silver Nanoparticles., J. Phys. Chem. C, 116, 8305 (2012).
- Karen I. Peterson, .Measuring the Density of a Sugar Solution: A General Chemistry Experiment Using a Student-Prepared Unknown., J. Chem. Ed. 85, 1089 (2008).
- Karen I. Peterson, David Pullman,Wei Lin, Andrea J. Minei, and Stewart E. Novick, .Microwave spectra and ab initio studies of Ar-propane and Ne-propane complexes: structure and dynamics., J. Chem. Phys. 127, 184306 (2007).
- D. Pullman and K. I. Peterson, "Investigating Intermolecular Interactions via Scanning Tunneling Microscopy: An Experiment for the Physical Chemistry Laboratory", J. Chem. Ed. 81, 549 (2004)
