Thomas Cole
Associate Professor, Organic Chemistry
office: CSL 210A
phone: 619-594-5579
email: tcole@sdsu.edu

Cole Group Page
Research Interests
Our research interests are in the exploration and development of organometallic chemistry of the transmetallation of organic groups between metals and boron compounds. This research program is composed of two major projects: the migration of organic groups from organoboranes to metals, giving rise to new organometallic complexes based in part on groups formed using boranes and the transfer of organic groups from metals to boron, yielding new unavailable organoboranes. These projects show exceptional promise in advancing synthetic organic methodology combining the attributes of metals and organoboranes.
Organometallic compounds frequently have unique reactivities, selectively forming novel organic fragments that would be difficult or impossible to prepare using classical organic reactions. Unfortunately, these metals frequently have limited carbon-carbon bond forming reactions. Organoboranes have well established high yielding carbon-carbon bond forming reactions. The migration of an organic group from a metal to boron would permit new applications of these novel organic fragments to synthesis, using the organoboranes as intermediates. We have found that alkenyl groups can be transferred with retention of stereochemistry to a variety of organohaloboranes. This reaction appears to be a general route to vinyl boranes with equals or surpasses regioselectivity of hydroboration methods. In contrast to the alkenyl groups, alkyl zirconium complexes undergo a facile isomerization as shown below. These alkyl groups can also be transferred to haloboranes forming a variety of structurally different organohalo- and organoboranes.
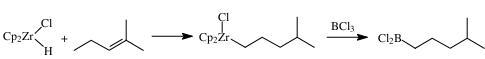
This transmetallation reaction gives a new route to these primary alkyboranes with very high regioselectivity that would be difficult to prepare using previously established routes. This route has recently been used to prepare borolanes and explore their application to other boranes and synthesis.
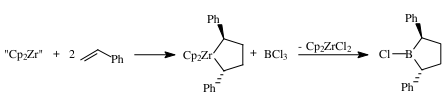
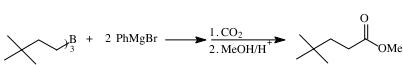
Research in progress has demonstrated another type of transmetallation, formation of Grignard reagents for organoboranes. We have found that primary alkyl groups migrate from organoboranes to the magnesium bromide cation in yields comparable to Grignard reagents prepared conventionally. This reaction allows one to potentially take advantage of the selectivity of hydroboration and organoborane reactions to form specific organic fragments. This approach is expected to form Grignard reagents that cannot be readily prepared using conventional methods.
Selected Publications
- "Preparation of alkyltrifluoroborates via Matteson Hydroboration Method,"
Zillman, David J.; Cole, Thomas E.,
J. Organomet. Chem. 865, 159-165 (2018). (doi: 10.1016/j.jorganchem.2018.03.027.) - "Reductive alkylation of p-benzoquinone using mixed organoboranes,"
Zillman D. J., Hincapie G. C., Savari M. R., et al.,
Tetrahedron Lett. 51, 3033-3036 (2010). - "New C-2-symmetric bis(sulfonamide)-cyclohexane-1,2-diamine-RhCp* complex and its application in the asymmetric transfer hydrogenation (ATH) of ketones in water,"
Cortez N. A., Rodriguez-Apodaca R., Aguirre G, et al.,
Tetrahedron Lett. 47, 8515-8518 (2006).
