
Gu Group Page
Curriculum Vitae
- Professor, San Diego State University, 2025-present
- Associate Professor, San Diego State University, 2022-2025
- Assistant Professor, San Diego State University, 2016-2022
- Postdoctoral Fellow, National Renewable Energy Lab, 2013-2016
- Postdoctoral Research Associate, Princeton University, 2011-2013
- PhD, Tulane University, 2006-2011
Research Interests
The growing need for clean and renewable energy has motivated the development of deployable systems to harvest and store earth's abundant solar energy resources. One promising approach is to collect and store solar energy within chemical bonds by forming fuels such as hydrogen and alcohols (solar fuels). This approach is similar to that of the natural photosynthestic process and thus has been dubbed "artificial photosynthesis." An example of this process is by utilizing the semiconductor as both the light absorber as well as the catalyst to split water into H2 and O2. The ultimate goal of these efforts is to develop a standalone photoelectrochemical cell in which water reduction and oxidation reactions occur efficiently under photo induced bias (ideally 1 sun), precluding the introduction of external electricity. In order to achieve this goal, the integration of stable light absorbing materials and effective catalysts to enhance the interfacial kinetics and fundamental mechanistic understanding of this coupled water splitting system still necessitate further explorations. In my lab, the majority of research focuses on two areas of solar energy conversion described below.
Development of Light Absorber-Catalyst Artificial Photosynthesis Assembles
Semiconductor electrodes that have been modified by molecular catalysts have been investigated extensively with various polymeric backbone attachments. However, few studies have been focused on the direct attachment of molecular catalysts onto semiconductor surfaces. By carefully adjusting the energetics between semiconductor and molecular catalyst interface, the efficiency of energy extraction from the photogenerated electron-hole pairs can be optimized. Adding an active catalyst unit to the surface of semiconductor is a feasible strategy to both improve the stability and maintain catalytic activity of a photoelectrochemical device. In a previous study we showed that by using an atomic layer deposited TiO2 layer as well as an earth abundant molecular catalyst we were able to protect the GaInP2 surface.

Figure 1. Representation of a photocathode where the GaInP2 semiconductor has been coated with TiO2. This surface modification allows a cobaloxime molecular catalyst to be bonded to the semiconductor
In the natural photosynthetic process, chemical energy is generated by light conversion followed by proton and electron transfer events (or proton coupled electron process). This process typically begins with light being absorbed by a harvesting pigment. The energy generated by light absorption is then transferred through different chromophores via energy cascade and eventually reaches a reaction center. At the reaction center, the light energy is converted to chemical energy through a multi-electron, multi proton transfer reactions. The basis of artificial photosynthesis is to mimic the photosynthetic process using semiconductors and molecular catalyst assemblies. Through an improved understanding of the coupled electron and energy transfer process in design assemblies could provide significant information for the future design and implementation of industrial scale artificial photosynthesis systems.
Understanding the Electron and Energy Transfer Mechanism in Light Harvesting Systems
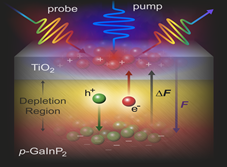
Figure 2. Schematic of the transient photoreflectance measurement. A pump pulse modulates the reflectance of a broadband pulse and creates free charge carriers within the depletion region of a semiconductor. The technique helps us understand the semiconductor and protection layer charge transfer kinetic.
Graduate students who work in my lab will receive an interdisciplinary training in inorganic chemistry (i.e. organometallic ligand and catalyst development, solid state chemistry), physical chemistry (i.e. nanosecond charge transfer and surface chemistry) and material science (i.e. surface characterization and material chemistry). State-of-the-art analytical techniques such as NMR spectroscopy, IR, fluorimetry, and ICP-MS will be used to shine light on the reaction mechanism. SEM, TEM and XPS will help us to monitor the morphology change for the material interface. In addition, the interactive nature of our research would provide us various collaborative opportunities between departments, schools and national labs, resulting in excellent learning opportunities for the graduate students. We always welcome graduate students and postdocs who are talented, determined and responsible to join our lab and together we will work towards finding alternative solutions to solve our energy crisis and environmental issues.
Recent Publications
- "Enhancing CO2 Reduction Efficiency on Cobalt Phthalocyanine via Axial Ligation,"
Kang H., Staples-West A., Washington A., Turchiano C., Cooksy A., Huang J., Gu J.,
Chem. Cat. Chem. 15, e202300576 (2023). (doi: 10.1002/cctc.202300576.) - "Selective NOx- Electroreduction to Ammonia on Isolated Ru Sites,"
Ke Z., He D., Yan X., Hu W., Williams N., Kang H., Pan X., Huang J., Gu J., Xiao X.,
ACS Nano 17, 3483-3491 (2023). (doi: 10.1021/acsnano.2c09691.) - "Surface Reorganization of Transition Metal Dichalcogenide Nanoflowers for Efficient Electrochemical Coenzyme Regeneration,"
Williams N., Hahn K., Goodman R., Chen X., Gu J.,
ACS Applied Materials and Interfaces 15, 3925-3933 (2023). (doi: 10.1021/acsami.2c17483.) - "Zinc Single Atom Confinement Effects on Catalysis in 1T-Phase Molybdenum Disulfide,"
Younan S.M., Li Z., Yan X., He D., Hu W., Demetrashvili N., Trulson G., Washington A., Xiao X., Pan X., Huang J., Gu J.,
ACS Nano (2022). (doi: 10.1021/acsnano.2c09918.) - "Terahertz Conductivity of Semiconducting 2H and Metallic 1T Phases of Molybdenum Disulfide,"
Capobianco M.D., Younan S.M., Tayvah U., Pattengale B., Neu J., Gu J., Brudvig G.W.,
Journal of Physical Chemistry Letters 13, 8319-8326 (2022). (doi: 10.1021/acs.jpclett.2c02283.) - "Atomically Interfacial Engineering on Molybdenum Nitride Quantum Dots Decorated N-doped Graphene for High-Rate and Stable Alkaline Hydrogen Production,"
Huang Y., Zhou W., Kong W., Chen L., Lu X., Cai H., Yuan Y., Zhao L., Jiang Y., Li H., Wang L., Wang L., Wang H., Zhang J., Gu J., Fan Z.,
Advanced Science 9, 2204949 (2022). (doi: 10.1002/advs.202204949.) - "Manipulating Interfacial Dissolution-Redeposition Dynamics to Resynthesize Electrode Surface Chemistry,"
Hu A., Zhang Y., Yang F., Hwang S., Sainio S., Nordlund D., Maxey E., Dai Q., Gu J., Li L., Lin F.,
ACS Energy Letters 7, 2588-2594 (2022). (doi: 10.1021/acsenergylett.2c01186.) - "Distance dependent energy transfer dynamics from a molecular donor to a zeolitic imidazolate framework acceptor,"
Hu W., Yang F., Pietraszak N., Gu J., Huang J.,
Physical Chemistry Chemical Physics 22, 25445-25449 (2020). (doi: 10.1039/d0cp03995k.) - "Manipulating Coordination Structures of Mixed-Valence Copper Single Atoms on 1T-MoS2for Efficient Hydrogen Evolution,"
Li Z., Yan X., He D., Hu W., Younan S., Ke Z., Patrick M., Xiao X., Huang J., Wu H., Pan X., Gu J.,
ACS Catalysis 12, 7687-7695 (2022). (doi: 10.1021/acscatal.2c00759.) - "Structure-Induced Stability in Sinuous Black Silicon for Enhanced Hydrogen Evolution Reaction Performance,"
Patrick M., Yang F., Vakki W., Aguiar J.A., Gu J.,
Advanced Functional Materials 31, 2008888 (2021). (doi: 10.1002/adfm.202008888.) - "Solar-assisted co-electrolysis of glycerol and water for concurrent production of formic acid and hydrogen,"
Ke Z., Williams N., Yan X., Younan S., He D., Song X., Pan X., Xiao X., Gu J.,
Journal of Materials Chemistry A 9, 19975-19983 (2021). (doi: 10.1039/d1ta02654b.) - "Graphitic-N highly doped graphene-like carbon: A superior metal-free catalyst for efficient reduction of CO2,"
Li J., Zan W.-Y., Kang H., Dong Z., Zhang X., Lin Y., Mu Y.-W., Zhang F., Zhang X.-M., Gu J.,
Applied Catalysis B: Environmental 298, 120510 (2021). (doi: 10.1016/j.apcatb.2021.120510.) - "Open and close-ended CoMoS3 nanotubes for hydrogen evolution in acidic and basic conditions,"
Li Y., Hou X., Gu J., Mikhaylova V., Chen K., Zhang H., Han S.,
Journal of Energy Chemistry 57, 34-40 (2021). (doi: 10.1016/j.jechem.2020.08.068.) - "Guanidyl-implanted UiO-66 as an efficient catalyst for the enhanced conversion of carbon dioxide into cyclic carbonates,"
Gao A., Li F., Xu Z., Ji C., Gu J., Zhou Y.-H.,
Dalton Transactions 51, 2567-2576 (2022). (doi: 10.1039/d1dt04110j.) - "Dynamic Evolution and Reversibility of Single-atom Ni(II) Active Site in 1T-MoS2 electrocatalysts for hydrogen evolution,"
Pattengale, B; Huang, Y.; Yan, X.; Yang, S.; Younan, S. ; Hu, W.; Li, Z.; Lee, S.; Pan, X.; Gu, J.*; Huang, J.*,
Nature. Commun. 11, 4114 (2020). (doi: 10.1038/s41467-020-17904-z.) - "Size-dependent Ni-based electrocatalysts for selective CO2 reduction,"
Li, Z.; He, D.; Yan, X.; Dai, S.; Younan, S.; Ke, Z.; Pan, X.; Xiao, X.; Wu, H.*, Gu,J.*,
Angew. Chem. Int. Ed. (2020). (doi: 10.1002/ange.202000318R1.) - "Photo/Bio-Electrochemical Systems for Environmental Remediation and Energy Harvesting,"
Fan Yang, Zunjian Ke, Zhida Li, Margaret Patrick, Zeinab Abboud, Nobuyuki Yamamoto, Xiangheng Xiao, Jing Gu,
Chem. Europe 13, 3391-3403 Review (2020). (10.1002/cssc.202000203.) - "Polyoxometalate-Based Photoactive Hybrid: Uncover the First Crystal Structure of Covalently Linked Hexavanadate-Porphyrin Molecule,"
Yingting Zhu, Yichao Huang, Qi Li, Dejin Zang, Jing Gu, Yajie Tang, and Yongge Wei,
Inorg. Chem. 59, 2575-2583 (2020). (10.1021/acs.inorgchem.9b03540.) - "Tuning Internal Strain in Metal—Organic Frameworks via Vapor Phase Infiltration for CO2 Reduction,"
Jing Gu, Fan Yang, Wenhui Hu, Chongqing Yang, Margaret Patrick, Andrew L. Cooksy, Jian Zhang, Jeffery A. Aguiar, Cheng-cheng Fang, Yinghua Zhou, Ying Shirley Meng, Jier Huang,
Angew. Chem./Angew. Chem. Int. Ed. 59/132, 4572-4580/4602-4610 (2020). (doi: 10.1002/anie.202000022.)
